Kai Siegbahn and colleagues from Uppsala University in Sweden recognized the potential of using photoelectrons for chemical analysis (thus, Electron Spectroscopy for Chemical Analysis) and was awarded the Nobel Prize for Physics in 1981 for these contributions. Where E kinetic is the kinetic energy of the photoelectron measured by the instrument, E photon is the energy of the incident photon (X-ray in this case, which is a known and fixed value), E binding is the binding energy of a given electron, and φ is the work function, the energy difference between the vacuum energy (Ev) level and the Fermi (Ef) level of a solid.ĭr. These relations are represented by the equation: E kinetic = E photon ( ℎ ν) - E binding - φ The kinetic energy of an emitted electron is related to the binding energy of each electron, and because atoms have multiple orbitals at different energy states, the resulting response will be a range of emitted electrons with different binding energies (and kinetic energies) thus producing an XPS spectrum. Einstein predicted that photoelectrons would be produced from a material when the energy of impinging photons exceed the binding energy of electrons in that material the energy is proportional to the frequency ( ℎ ν ) not the intensity or duration of exposure to the incident electromagnetic radiation.

XPS is an application of the photoelectric effect (Acrobat (PDF) 184kB Jul29 21) described by Einstein (1905, and was awarded the Nobel Prize in 1921), in which electrons are emitted from atoms in response to impinging electromagnetic radiation.
Palladium asymmetric xps peak license#
Reuse: This item is offered under a Creative Commons Attribution-NonCommercial-ShareAlike license You may reuse this item for non-commercial purposes as long as you provide attribution and offer any derivative works under a similar license.
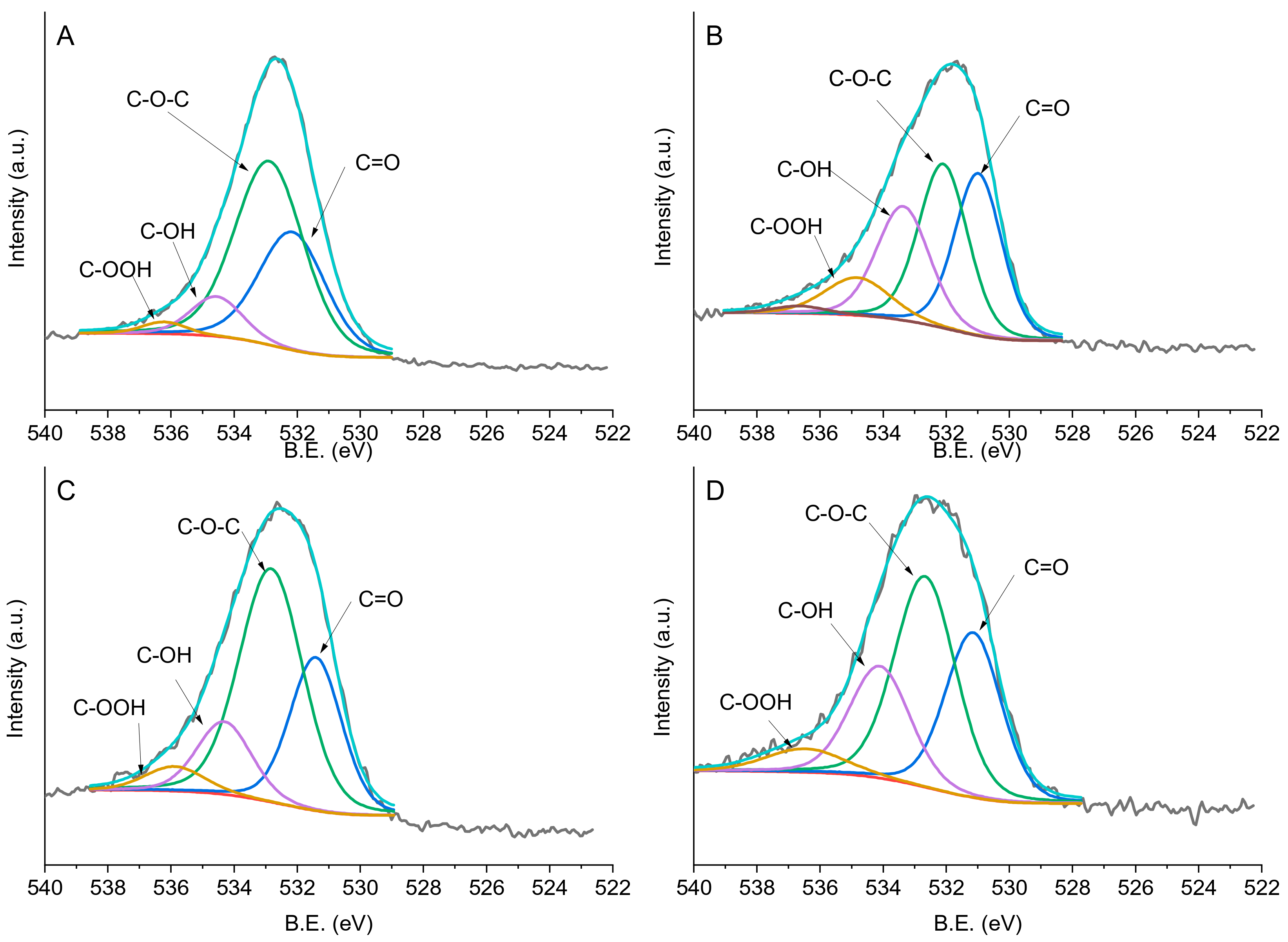
It is almost always the case that the surface composition and chemistry of materials, measured on the order of a few atomic layers (~10 nm), is different from the "bulk" composition determined by methods such as energy dispersive spectrometry (EDS) with excitation volumes that can extend as much as 3 microns into the material.įundamental Principles of X-Ray Photoelectron Spectroscopy XPS is used to support research on surface-mediated processes such as sorption, catalysis, redox, dissolution/precipitation, corrosion, and evaporation/deposition type reactions. XPS is used to characterize the surfaces of diverse materials such as inorganic compounds (minerals), semiconductors, organic compounds, and thin films and coatings on natural and engineered materials. XPS is routinely used to determine a) the composition of material surfaces (elemental identification), the relative abundances of these components on surfaces (semi-quantitative analysis), and c) the chemical state of polyvalent ions by measuring the binding energies of elements, which is related to the nature and strength of their chemical bonds.
Palladium asymmetric xps peak free#
The element can be found as a free metal and alloyed within platinum and gold in deposits located in Ethiopia, South and North America and the Ural Mountains.X-ray photoelectron spectroscopy (XPS) is a surface sensitive, non-destructive technique used routinely to analyze the outermost ~10 nm (~30 atomic layers) of natural and engineered materials. Palladium is commercially produced from nickel-copper deposits. Palladium was named after the asteroid Pallas, which was discovered two years prior to the element’s discovery. In 1803, palladium was first discovered in crude platinum ore from South America by W. When dissolved in hydrochloric acid, palladium absorbs up to 900 times its own volume of hydrogen at room temperature. Some notable features of palla- dium include its tarnish resistance, low density and melting point, and its chemical attraction to sulfuric and nitric acid. Primarily used as an industrial catalyst and in jewelry, palladium is a rare silver-white transition metal similar to platinum. Obtained From: platinum, nickel, copper, mercury ores
Pd3d region has well separated spin-orbit components (Δ=5.26 eV).

